You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
Lead-free bullet casting alloy
- Thread starter Ian
- Start date
Ian
Notorious member
I think I figured it out, that ternary system describes the liquid phase only at different temperatures (liquidus point), but does not include any solidus or L+alpha or L+Beta phases. If .5 Sb in that system isn't forming Sb/Sn, then the Sb must be free in the solution? IDK, whatever it does, the Sb seems to be helping ductility and isn't otherwise causing problems so unless anyone has better ideas, this is probably good 'nuff.
Lookie what I found last night! It was near the right thigh bone in the quad muscle where it bounced off the loin hide (leaving the gas check very stuck there) and went back into the leg.

This is bullet #13 ever fired from this alloy, first two went into the trap, five for a group at 50, next five at 100, then this one for venison. Pretty short test run.
Lookie what I found last night! It was near the right thigh bone in the quad muscle where it bounced off the loin hide (leaving the gas check very stuck there) and went back into the leg.

This is bullet #13 ever fired from this alloy, first two went into the trap, five for a group at 50, next five at 100, then this one for venison. Pretty short test run.
fiver
Well-Known Member
I was half thinking the bullet started tumbling when it went through the heart.
if it sheared the nose on a rib or something that would make sense, but the hole going in is what give me the idea, of course just punching the skin could have set it tumbling too at the angle you took the shot at.
It however was also occurring to me you started sheared the nose somewhere and hitting the solid muscle [heart] finished the job, and/but you had to have enough mass pushing things along to get to the off side.
if it sheared the nose on a rib or something that would make sense, but the hole going in is what give me the idea, of course just punching the skin could have set it tumbling too at the angle you took the shot at.
It however was also occurring to me you started sheared the nose somewhere and hitting the solid muscle [heart] finished the job, and/but you had to have enough mass pushing things along to get to the off side.
fiver
Well-Known Member
referring back to the chart in post 118.
question.
am I seeing a line up in the right hand corner there referring to a Bi alloy but with a SbSn chain.
if so it's showing a slush phase? but I'm not sure at what point it shows a solid phase nor am I sure if that's supposed to be an equal chain length.
it kind of occurs to me it might be beneficial, if that's what I'm seeing,, a lower 3/3 type alloy might be worth looking into.
anyhow.
speculation because I got nuthin in hand to look at.
the higher operating temp is what drew my eye in that direction, but I don't know where the lines go in and out of the chart right there, or why it would be such a small window.
unless that chain is just happening during the liquid phase, but being broken as the temp comes down into the solid [as they look for a place to go] and once again if so that would explain why the stuff is trying to squirt out the sides like I mentioned before.
the more I look at the charts the more I see predictions being correct, but also sort of some contradictions to what the charts are showing,,, all basically depending on the temperature of the alloy.
I guess what I'm saying here is the alloy temperature is gonna determine/influence the internal structure of the alloy and the casting outcome more than the mold temperature [within reason] here.
question.
am I seeing a line up in the right hand corner there referring to a Bi alloy but with a SbSn chain.
if so it's showing a slush phase? but I'm not sure at what point it shows a solid phase nor am I sure if that's supposed to be an equal chain length.
it kind of occurs to me it might be beneficial, if that's what I'm seeing,, a lower 3/3 type alloy might be worth looking into.
anyhow.
speculation because I got nuthin in hand to look at.
the higher operating temp is what drew my eye in that direction, but I don't know where the lines go in and out of the chart right there, or why it would be such a small window.
unless that chain is just happening during the liquid phase, but being broken as the temp comes down into the solid [as they look for a place to go] and once again if so that would explain why the stuff is trying to squirt out the sides like I mentioned before.
the more I look at the charts the more I see predictions being correct, but also sort of some contradictions to what the charts are showing,,, all basically depending on the temperature of the alloy.
I guess what I'm saying here is the alloy temperature is gonna determine/influence the internal structure of the alloy and the casting outcome more than the mold temperature [within reason] here.
Last edited:
Ian
Notorious member
I think you're right about the bullet being "pre cracked" after it hit the rib and the heart being the "straw" that made it come apart. I also cannot be certain that the nose didn't blow off in the rib and the angled fracture start it tumbling so it hit the heart sideways (the entrance hole in the heart is kind of elliptical) and the sideways hit blew the exit side apart like a totten tomato. There were three holes in the diaphragm and the liver was definitely hit but it honestly I didn't study the holes very well to see what shape(s) of things made them. The chest cavity was full of sand-sized bone fragments and a 2" chunk of rib was missing. What surprised me was how straight the bullet went and how much bone/organs/meat it took to stop it.
When you add more than one percent antimony, I think it starts doing weird stuff that actually makes the alloy MORE brittle, but I'm only basing that on the 57% solder that was tested. With something like 3/3 or 5/5 it might be a different story. My problem is not having a ternary phase diagram of the actual alloy I was using to see what is going on.
When you add more than one percent antimony, I think it starts doing weird stuff that actually makes the alloy MORE brittle, but I'm only basing that on the 57% solder that was tested. With something like 3/3 or 5/5 it might be a different story. My problem is not having a ternary phase diagram of the actual alloy I was using to see what is going on.
Your 88/12/0.75 puts it right at the edge of the BCT_A5 - sbsn curve. The BCT_A5 is a funny cubic tin structure that I assume causes some fine grain structure. Possibly it locks up some tin so tin 'blobs' don't form. SbSn from what I can find is an intemetallic that decomposes readily as it cools, sometimes. Even if all the Sb combines with Sn, still only 0.75%?
My understanding of this grain refining is the Bi & Sn are an elongated rectangle (rhomboid) jumble with course grain structure. Adding the Sb causes tin to form a nice cube that separates the jumble.
Pb on the other hand, is a nice cube structure (rubik cube form) and is malleable as the layers of the cube slide easily. We put other stuff in it (to harden) in lumps that replace some of the nice cubes and increase the force required to slide.
So for Bi we want to do the opposite to increase malleability. Additionally, Bi is diamagnetic, meaning it doesn't even stick to itself very well - and is very crumbly. So we add tin to glue it together. But we have to add something to the tin so it doesn't cold flow into blobs. Thus the Sb.
Your bullet has a large grain struct where it broke -at the groove as normal - interested if all that bullet structure is the same? Why do I ask? Pouring lead, nose cools fast first. Possibly a change in grain due to slower cooling and precipitation flow during the slow cool? Or pressure generated heat during impact. Just guesses at this point.
I am NOT a metallurgist but did take a course in strength of materials with a research prof (guy who 'invented' glass reinforced re-bar, very interesting story).
My understanding of this grain refining is the Bi & Sn are an elongated rectangle (rhomboid) jumble with course grain structure. Adding the Sb causes tin to form a nice cube that separates the jumble.
Pb on the other hand, is a nice cube structure (rubik cube form) and is malleable as the layers of the cube slide easily. We put other stuff in it (to harden) in lumps that replace some of the nice cubes and increase the force required to slide.
So for Bi we want to do the opposite to increase malleability. Additionally, Bi is diamagnetic, meaning it doesn't even stick to itself very well - and is very crumbly. So we add tin to glue it together. But we have to add something to the tin so it doesn't cold flow into blobs. Thus the Sb.
Your bullet has a large grain struct where it broke -at the groove as normal - interested if all that bullet structure is the same? Why do I ask? Pouring lead, nose cools fast first. Possibly a change in grain due to slower cooling and precipitation flow during the slow cool? Or pressure generated heat during impact. Just guesses at this point.
I am NOT a metallurgist but did take a course in strength of materials with a research prof (guy who 'invented' glass reinforced re-bar, very interesting story).
Ian
Notorious member
That's a pretty good explanation, Popper.
I took a materials course as an ME and it was heavy on phase diagrams but also heavy on ferrous materials and binary systems. That was 25 years ago. The part about how Alpha or Beta forms with similarly structured components of a system makes it seem that Bi wouldn't form a true solution with anything. I also remember calculating the percentages of alpha/beta + L and something about that curve intersecting the eutectic line and how everything freezing after that was a eutectic mix of alpha and beta phases (coring).
If that is the case, perhaps the last part of this alloy to freeze is a small amount of 61/39 eutectic Bi/Sn? That would explain the main alloy freezing solid at about 400 and a small amount of liquid remaining until it cools at least another 100 to 150⁰.
I took a materials course as an ME and it was heavy on phase diagrams but also heavy on ferrous materials and binary systems. That was 25 years ago. The part about how Alpha or Beta forms with similarly structured components of a system makes it seem that Bi wouldn't form a true solution with anything. I also remember calculating the percentages of alpha/beta + L and something about that curve intersecting the eutectic line and how everything freezing after that was a eutectic mix of alpha and beta phases (coring).
If that is the case, perhaps the last part of this alloy to freeze is a small amount of 61/39 eutectic Bi/Sn? That would explain the main alloy freezing solid at about 400 and a small amount of liquid remaining until it cools at least another 100 to 150⁰.
fiver
Well-Known Member
so the general thought is that with the antimony we basically have a bullet with lets call it a 2 part construction. [inner core and outer shell]
1/2% can only influence so much of that tin.
if it is pulled to the outer part [and basically set free] because of the cooling speed and you have that internal L stage still going on then I can see how you could have 2 different grain structures.
maybe I'm seeing it wrong because you 'should' have a SbSn chain within the liquid but not within the cooled alloy according to that chart.
what we then see popping [sweating] out is free tin.
same as you see if you heat a PB alloy bullet with a torch [or just on top of your pot sometimes] it will start to sweat out beads of tin before the entire alloy melts.
only this is in reverse.
maybe it is the bismuth and not the tin because it does have a lower melt point and the tin is locked up [blocked] by the Sb addition like Popper explains above.
that doesn't seem right, though it sort of should given the solid and melted temps, but LOL lot of them Buts...
when Ian added more tin the melt point went down.
[that to me is like adding Carnuba to bees wax and lowering the melt point to like 130-F]
I guess the only real way to know is to microscope the escaping alloy or XRF the little pieces saved.
1/2% can only influence so much of that tin.
if it is pulled to the outer part [and basically set free] because of the cooling speed and you have that internal L stage still going on then I can see how you could have 2 different grain structures.
maybe I'm seeing it wrong because you 'should' have a SbSn chain within the liquid but not within the cooled alloy according to that chart.
what we then see popping [sweating] out is free tin.
same as you see if you heat a PB alloy bullet with a torch [or just on top of your pot sometimes] it will start to sweat out beads of tin before the entire alloy melts.
only this is in reverse.
maybe it is the bismuth and not the tin because it does have a lower melt point and the tin is locked up [blocked] by the Sb addition like Popper explains above.
that doesn't seem right, though it sort of should given the solid and melted temps, but LOL lot of them Buts...
when Ian added more tin the melt point went down.
[that to me is like adding Carnuba to bees wax and lowering the melt point to like 130-F]
I guess the only real way to know is to microscope the escaping alloy or XRF the little pieces saved.
Ian
Notorious member
It isn't pure tin leaking out of the Bi alloy because it is way below the melt point of tin. I think it has to be a eutectic mixture because that's what freezes last accirding to the binary phase chart.
Popper's chart shows only the liquid solution phase of the ternary mix, and the SbSn seems to exist at certain concentrations of all three, indeoende t of temperature as long as the system is warm enough for all to be fully melted. What we don't see there is the solidus phases beginning as it cools. That is what we need and rhe closest we have is the solder phase chart I posted previously, together with the Bi/Sn binary phase chart. The two are so different it's tough to tell what's going on with this ternary bullet metal.
Popper's chart shows only the liquid solution phase of the ternary mix, and the SbSn seems to exist at certain concentrations of all three, indeoende t of temperature as long as the system is warm enough for all to be fully melted. What we don't see there is the solidus phases beginning as it cools. That is what we need and rhe closest we have is the solder phase chart I posted previously, together with the Bi/Sn binary phase chart. The two are so different it's tough to tell what's going on with this ternary bullet metal.
Interesting https://link.springer.com/article/10.1007/BF02665790.
Cu additon as well as Iodine look like fun.
Cu additon as well as Iodine look like fun.
fiver
Well-Known Member
well..
I looked and to me it's showing about 1% sulpher makes all kinds of changes 2% seems to overlap but spreads out more.
you could pinpoint those changes with a 0.1% change under 1%
the way they showed getting it in appears to be by melting it and pouring it right in the melted Bi.
don't know why I looked I know I'm gonna get a headache, but I couldn't stop myself.
I looked and to me it's showing about 1% sulpher makes all kinds of changes 2% seems to overlap but spreads out more.
you could pinpoint those changes with a 0.1% change under 1%
the way they showed getting it in appears to be by melting it and pouring it right in the melted Bi.
don't know why I looked I know I'm gonna get a headache, but I couldn't stop myself.
Ian
Notorious member
More shooting today to make sure it wasn't a fluke. Dirty rifle from last session. Due to LBT lube throwing awful flyers I reduced the lube to just the space above the gas check and the next groove above it and loaded one fouler with a wart on its nose. Shoulda loaded three foulers, oh well. It shoots once the sludge is limbered up.

I might mention again that this rifle hasn't managed to shoot an inch group or less for ten with anything, including well-assembled match jacketed loads. 1.5" at 100 has been the average for HV cast.
One observation that some might find of interest is this relatively mild load didn't get the barrel hot. It was just warm to the touch after launching 11 rounds in about six minutes.

I might mention again that this rifle hasn't managed to shoot an inch group or less for ten with anything, including well-assembled match jacketed loads. 1.5" at 100 has been the average for HV cast.
One observation that some might find of interest is this relatively mild load didn't get the barrel hot. It was just warm to the touch after launching 11 rounds in about six minutes.
fiver
Well-Known Member
I'm not Ian, but I'd say it's a bit on the soft side for his conditions.
you know Ian that poor rifle is so close to wanting to shoot.
it's at least predictable.
have you tried breaking the barrel loose and re-torqueing it?
after all the other stuff you done to it I'm coming to the conclusion the barrel and receiver just ain't connected well.
you know Ian that poor rifle is so close to wanting to shoot.
it's at least predictable.
have you tried breaking the barrel loose and re-torqueing it?
after all the other stuff you done to it I'm coming to the conclusion the barrel and receiver just ain't connected well.
Ian
Notorious member
The problem with the lube is it's too stiff and doesn't flow until the barrel is warm to the touch. This causes two flyers from a clean barrel and three from a fouled one. When the barrel cools off it is almost impossible to get a tight patch through the bore because of the heavy deposit of viscous carbon/lube goo.
You know, I actually thought about the barrel interface last week. Been thinking about setting back just a couple threads and rechambering to get rid of that looooonnng throat and let me fit bullets to the throat and still allow the cartriges in the magazine. I wanted to see how well the barrel and receiver fit, how tight the nut was, and if there was any threadlocker there to soak up vibration. The barrel acts loose, like it's micro-fretting on the receiver threads. Harold Vaughn did some pretty exhaustive studies of rifle barrel threaded connections and found lots of issues there which he addressed in a variety of ways. It almost became the limiting factor for accuracy in his .270 before he figured out a system of clamping and torque that worked. He couldn't find a way to make the thread clamp load higher than bolt thrust, so the barrel would worm around during each shot, so much so he was able to measure the shift with a test indicator.
The other thing I've been meaning to try is a shim at the front of the forearm.
You know, I actually thought about the barrel interface last week. Been thinking about setting back just a couple threads and rechambering to get rid of that looooonnng throat and let me fit bullets to the throat and still allow the cartriges in the magazine. I wanted to see how well the barrel and receiver fit, how tight the nut was, and if there was any threadlocker there to soak up vibration. The barrel acts loose, like it's micro-fretting on the receiver threads. Harold Vaughn did some pretty exhaustive studies of rifle barrel threaded connections and found lots of issues there which he addressed in a variety of ways. It almost became the limiting factor for accuracy in his .270 before he figured out a system of clamping and torque that worked. He couldn't find a way to make the thread clamp load higher than bolt thrust, so the barrel would worm around during each shot, so much so he was able to measure the shift with a test indicator.
The other thing I've been meaning to try is a shim at the front of the forearm.
Last edited:
fiver
Well-Known Member
I wonder if that shim trick actually forces the threads to bottom out and top out the other direction.
I have full length bedded a number of rifles with thinner barrels with very good success, I'm now wondering if I wasn't just bowing everything up putting the barrel under tension.
but it has worked on previously bedded rifles too.
I have full length bedded a number of rifles with thinner barrels with very good success, I'm now wondering if I wasn't just bowing everything up putting the barrel under tension.
but it has worked on previously bedded rifles too.
Ian
Notorious member
I would guess that depends on how much shim you used. This rifle's action is 100% stress-free pillar bedded now, but it has a 24" medium-countour barrel, muzzle brake, and a pretty heavy suppressor hanging off the end. The receiver diameter is quite small, so there is a tremendous amount of stress on the barrel tenon which is only increased in the same direction when the rifle is fired. Since the bedding is so good now and the laminated tacticool stock has a wide, heavy, almost BR type forearm, I think shimming, bedding a strip, or at least putting a strip of closed-cell foam to neutralize the sag and no more might be in order. I still want to yank the barrel off and have a look-see at what's going on there first. If all that doesn't help I still have a complete Tubbs lapping kit to try.
Last edited:
"The Bi1-xSb x single-crystal samples (0 ≤ x ≤ 0.17) used for ARPES experiments were each cleaved from a boule grown from a stoichiometric mixture of high-purity elements. The boule was cooled from 650 °C to 270 °C over a period of five days and was annealed for seven days at 270 °C. The samples naturally cleaved along the (111) plane, which resulted in shiny flat silver surfaces. (bold - sic.) X-ray diffraction measurements were used to check that the samples were single phase, and confirmed that the Bi0.9Sb0.1 single crystals presented in this paper have a rhombohedral A7 crystal structure (point group R
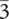 m), with room-temperature (300 K) lattice parameters a = 4.51 Å and c = 11.78 Å indexed using a rhombohedral unit cell. The X-ray diffraction patterns of the cleaved crystals exhibit only the (333), (666), and (999) peaks, showing that the cleaved surface is oriented along the trigonal (111) axis. Room-temperature data were recorded on a Bruker D8 diffractometer using Cu Kα radiation (λ = 1.54 Å) and a diffracted-beam monochromator. The in-plane crystal orientation was determined by Laue X-ray diffraction. During the angle-resolved photoemission spectroscopy (ARPES) measurements a fine alignment was achieved by carefully studying the band dispersions and Fermi surface symmetry as an internal check for crystal orientation. "
m), with room-temperature (300 K) lattice parameters a = 4.51 Å and c = 11.78 Å indexed using a rhombohedral unit cell. The X-ray diffraction patterns of the cleaved crystals exhibit only the (333), (666), and (999) peaks, showing that the cleaved surface is oriented along the trigonal (111) axis. Room-temperature data were recorded on a Bruker D8 diffractometer using Cu Kα radiation (λ = 1.54 Å) and a diffracted-beam monochromator. The in-plane crystal orientation was determined by Laue X-ray diffraction. During the angle-resolved photoemission spectroscopy (ARPES) measurements a fine alignment was achieved by carefully studying the band dispersions and Fermi surface symmetry as an internal check for crystal orientation. "
Hsieh, D., Qian, D., Wray, L. et al. A topological Dirac insulator in a quantum spin Hall phase. Nature 452, 970–974 (2008) doi:10.1038/nature06843
So Bi0.9Sb0.1 does exist at room temp. Bi has different surface structure than bulk (center). Pressure (expansion due to being in a containing mould) does change the structure a bit. The graph I posted did show a solid of Sb2Sn3.
As the Fiver's 'coring' theory, maybe? But to have Sn,SbSn, BiSb,etc primarily at the surface, something besides cooling must cause migration of molecules in the liquid. Some work has been done in solidus in a magnetic field. Yea, right.
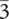
Hsieh, D., Qian, D., Wray, L. et al. A topological Dirac insulator in a quantum spin Hall phase. Nature 452, 970–974 (2008) doi:10.1038/nature06843
So Bi0.9Sb0.1 does exist at room temp. Bi has different surface structure than bulk (center). Pressure (expansion due to being in a containing mould) does change the structure a bit. The graph I posted did show a solid of Sb2Sn3.
As the Fiver's 'coring' theory, maybe? But to have Sn,SbSn, BiSb,etc primarily at the surface, something besides cooling must cause migration of molecules in the liquid. Some work has been done in solidus in a magnetic field. Yea, right.


